Forms of Macular Degeneration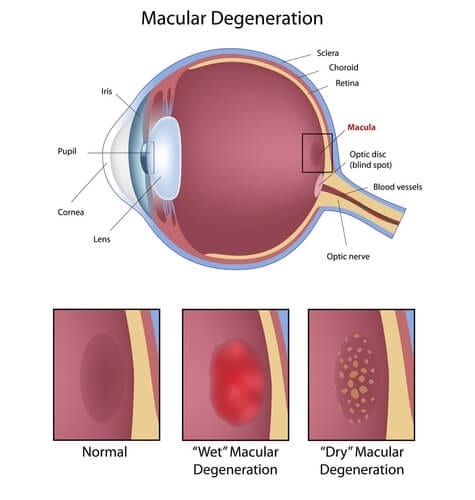
Macular Degeneration and Age-Related Macular Degeneration occurs in two main forms: a “dry” or atrophic type and a “wet” or exudative type. Everyone with macular degeneration initially has the dry form and every person with macular degeneration develops the disease in both eyes, however, one eye can worsen more quickly than the other. The dry form of AMD is NOT one disease; there are four major groups of dry AMD. The first three are classified by the size of the small yellow spots (called drusen) that develop in the layer of cells underneath the retina. Drusen come in three sizes: small, medium and large. Patients with small drusen have an excellent chance of keeping good vision during their lifetime. Patients with medium or large drusen have a much higher chance of developing the wet form of macular degeneration, especially if their drusen are associated with small clumps of brown pigment. Patients with medium or large drusen are considered to have “HIGH-RISK” dry AMD. The fourth type of dry macular degeneration is called geographic atrophy, in which the cells underneath the retina slowly wither away. Geographic atrophy only rarely changes into wet macular degeneration, however, it can cause a person to become legally blind if the disease affects the cells underneath the very center of the macula.
Preventive Measures for Macular Degeneration
Treatment for dry macular degeneration depends upon the type of disease present in a patient. All patients with dry macular degeneration should have a coating on the lenses of their eyeglasses to block the ultraviolet rays from the sun. Sunglasses are helpful when outdoors. A daily multivitamin with antioxidants, lutein, zeaxanthin, and zinc may slow the progression of high-risk dry AMD and wet AMD. We do not recommend taking more than 80 mg of zinc per day because this may be harmful to the retina and other organs.
 Diagnostics for Macular Degeneration
Diagnostics for Macular Degeneration
Although only 10% of patients with AMD will develop the wet type of degeneration, 90% of the patients with severe visual impairment have this form of the disease. In wet AMD, fluid builds up under the retina. Abnormal new blood vessels that grow like tiny weeds under the retina cause this fluid. The fluid can be seen during a retinal examination and documented with a noninvasive test called Ocular Coherence Tomography (OCT). The abnormal blood vessels and the leakage they cause are visualized using a safe intravenous dye test called a fluorescein angiogram. Without treatment, the blood vessels will eventually form a scar and lead to permanent loss of the central vision.
Treatments for Macular Degeneration
Some patients with high-risk dry AMD may be eligible for a new laser treatment that can cause the medium and large drusen to disappear within 6 months. Recently the laser treatment has proven to have a significantly beneficial effect on vision in two different well-designed studies. It has yet to be determined from the studies whether the laser treatment will reduce the risk of a patient getting the wet form of AMD; however, this information should be available soon. The new laser treatment for high-risk dry AMD is painless, takes less than 5 minutes and is performed in the office. Approximately 20 very mild laser spots are placed in a semicircular grid pattern around the edge of the macula. Most patients do not even notice the laser spots after treatment; if they are noticeable, the spots usually fade away within 10 days. If both eyes require treatment for the laser, they are treated about a week apart. The patient is asked to return for an exam and fluorescein pictures at 3 months and 6 months after the treatment. If the drusen have not regressed sufficiently after 6 months, another laser treatment may be repeated on the opposite side of the macula.
Every patient with dry macular degeneration is given a special chart with a checkerboard grid pattern called an Amsler Grid. The Amsler Grid that your Retina Specialist gave you has the instructions printed on the back. It is crucial for a patient with macular degeneration to look at his/her grid every day. The proper way to look at the grid is to hold it at a reading distance (with your reading glasses on) and cover one eye at a time. While looking at the round dot in the center, you’re verifying that all the lines appear straight. Some patients with dry macular degeneration may have a few wavy lines on the Amsler Grid; as long as the appearance of the grid does not change from day to day, there is no cause for concern. However, if the lines suddenly become more distorted or lines that were previously straight become wavy, then call the office IMMEDIATELY. We will make every effort to see you in the office within 24 hours. It is very important to understand any sudden noticeable change in the appearance of the grid almost always means there is a problem in the macula and we consider it to be an emergency.
Laser Therapy
Historically, the first widely used therapy for wet macular degeneration was thermal laser treatment, which has been performed for about 40 years. Because the laser energy is converted to heat (hence the name “thermal”), the major side effect of the laser treatment is a permanent dark or gray scar in the central area of vision. The rationale behind thermal laser treatment was to intervene as soon as possible after the new blood vessels form so the laser scar would be as small as possible. In the vast majority of patients, the vision did not improve or got worse after thermal laser treatment. A series of major studies performed in the 1980s and 1990’s called the Macular Photocoagulation Studies concluded the thermal laser treatment was beneficial in slowing down the anatomical progression of the wet macular degeneration and was better than the natural history, but again, the vast majority of patients almost inevitably continued to lose vision. Currently, thermal laser treatment is reserved for a very small percentage of patients with wet macular degeneration. Usually, it is used when the new blood vessels are well outside the center of the macula. Patients treated with the thermal laser for wet AMD have to be followed by a retinal specialist very carefully because the blood vessels can reappear almost two-thirds of the time and may need additional laser treatment.
Photodynamic Therapy (PDT) is a new laser treatment for wet macular degeneration that was approved by the Food and Drug Administration in 2000. This is a treatment for active wet macular degeneration. PDT is performed in the office by infusing an intravenous dye called Visudyne into a vein in the forearm for about 15 minutes. The dye collects in the abnormal new blood vessels growing under the macula. About 5 minutes after the dye has been given, a non-thermal (“cold”) laser light is focused onto the macula. The light creates a chemical reaction with the dye that causes the new blood vessels to clot. The most important advantage of Photodynamic Therapy is the blood vessels can be treated without damaging the retina since the laser does not produce heat. As a precaution, patients are asked to avoid direct sunlight for 48 hours after the treatment. Since the dye is activated by infrared light, which is found in sunlight, a person with the dye in his/her system can get a very severe sunburn until the Visudyne is cleared from the body. As with traditional laser treatment, patients must be monitored carefully following Photodynamic Therapy since the blood vessels often start to grow again and often require additional photodynamic laser treatment. The results of PDT as a treatment for wet AMD were initially promising but are now considered disappointing. It is most useful in only about 25% of patients with wet AMD and even then, only 6% of eyes improve 3 or more lines of vision after treatment. PDT may have a role in very few patients with wet AMD and current research is concentrating on the use of PDT in combination with other treatment methods.
VEGF Therapies
At this time, the unquestioned gold standard treatment for wet AMD is anti-vascular endothelial growth factor (VEGF) therapy, given as a series of injections into the vitreous cavity. VEGF is a family of 6 proteins produced all over the body, including in the eye that is critically important in the development of both normal and abnormal new blood vessels. In wet AMD, VEGF is produced in abnormally high amounts and leads to the formation of the abnormal new blood vessels that arise from the choroidal layer behind the retina. The new blood vessels are very leaky. VEGF also increases the leakage of fluid from these blood vessels into space underneath the retina and into the retina itself. Because the macula then looks “wet” when a retina specialist is examining the eye this is the derivation of the name: wet macular degeneration. The abnormal blood vessels are fragile and bleed easily resulting in hemorrhage in the macula in some patients with wet AMD.
All anti-VEGF treatments given today are administered as an injection into the vitreous cavity, which is a procedure performed in the office. The surface of the eye is numbed with drops and then an anesthetic injection of lidocaine is given under the conjunctiva (the “skin” covering the white part of the eye). The eye is then thoroughly cleaned and sterilized with betadine and antibiotic drops. The anti-VEGF medication is injected through the anesthetized area into the vitreous cavity using a very tiny needle. The procedure itself is painless and straightforward. However, it is considered an invasive procedure because we are entering the eye itself and there is a rare chance of complications such as cataract, retinal detachment, bleeding or infection. These complications occur in less than one thousand injections. Occasionally, the volume of medication injected temporarily interrupts the circulation to the eye and it might be necessary for the surgeon to remove some fluid from the front of the eye to restore the normal circulation. All patients receiving an injection are asked to take antibiotic drops for four days after the injection to lessen the chance of infection.
The most common question patients ask about any of the anti-VEGF agents is “when does the treatment stop”? The answer, like many things in medicine, is “it depends”. Some retina specialists will treat patients indefinitely because the clinical trials of these medications did just that. Some will treat until the lesion becomes inactive and then observe indefinitely for signs of recurrence. Some will treat until the lesion becomes inactive and then try to extend the time in-between injections. Others will try a combination of treatments – an anti-VEGF drug with a steroid injection or an anti-VEGF drug with PDT laser or all three. Still, others will start with Lucentis or Avastin until the lesion is dry and then perform “maintenance injections” with Macugen.
Macugen® Injections
In January 2005, Macugen became the first ant-VEGF drug approved by the Food and Drug Administration for the treatment of wet AMD. In the VISION clinical trial, about 6% of patients improved their visual acuity 3 or more lines and 90% experienced stabilization of vision. The medication is given every 6 weeks. In real-world clinical experience since 2005, Macugen probably is a little more effective than indicated by the study results, and most improvement occurs after 4 to 6 injections. The reason for this is most likely the VISION trial included many patients with the most severe forms of wet macular degeneration and very poor initial vision. When a subset of patients with less severe disease and better initial vision was analyzed, about 20% experienced 3 or more lines of vision gain. On the other hand, Macugen has proven to be an extraordinarily safe drug with no significant local or systemic side effects reported. We still do use Macugen in certain patients, such as those who have suffered a stroke within the previous 6 months or who have poorly controlled hypertension. In some instances, Macugen is used as “maintenance” therapy in patients whose wet macular degeneration has regressed using one of the other anti-VEGF treatments. Most retina specialists would agree Macugen is currently the third choice of the three available anti-VEGF agents for the vast majority of patients.
Avastin® Injections
In May 2005, Dr. Phil Rosenfeld of the Bascom Palmer Eye Institute reported the use of Avastin injections in the eye of a patient with wet macular degeneration. Avastin is an antibody against VEGF that was originally approved for intravenous use in patients with metastatic colon cancer. The reason we are allowed to treat patients with Avastin is a special circumstance referred to as “Off-Label”, in which a physician can use a drug approved for one disease in an unrelated disease if he believes it has the potential to benefit patients. Since 2005, the use of Avastin has become commonplace all over the world and is accepted by the retina specialist community as a standard of care. There are several clinical studies underway to determine Avastin’s effectiveness and safety, however, the results will not be known for several years. Avastin is most commonly given every 6 weeks, though treatment can vary according to the condition of the eye. Additionally, one of Avastin’s advantages is its low cost per dose (approximately $75) compared to Lucentis (approximately$2,500) and Macugen (approximately $1,000).
Lucentis® Injections
The FDA approved Lucentis for the treatment of wet AMD in July 2006. Lucentis is a very close relative of the Avastin molecule and is also an antibody to VEGF. According to the results of the ANCHOR and MARINA trials, Lucentis improved the vision 3 or more lines in about 40% of eyes and stabilized the vision by 95%. In terms of scientifically documented results, Lucentis is the best available drug currently approved for wet AMD. Although there has been some concern about potential systemic side effects including hypertension and stroke (for both Lucentis and Avastin), it is unclear at this point if these concerns are significant despite the hundreds of thousands of injections given worldwide. Lucentis is given every 4 to 8 weeks depending on the condition of the eye and the judgment of the retina specialist.
Eyelea® Injections
On November 18, 2011, The United States Food and Drug Administration (FDA) approved the use of Eylea (aflibercept) for treating wet age-related macular degeneration (AMD). Eylea, developed by Regeneron Pharmaceuticals, Inc. (Tarrytown, NY), is injected into the eye just like Avastin, Lucentis, and Macugen. Prior to its approval by the FDA, Eylea was known in the scientific literature as “VEGF Trap.” Eylea is a recombinant fusion protein (a “designer drug”) that binds to both VEGF (vascular endothelial growth factor) and PlGF (placental growth factor). VEGF and PlGF are proteins normally produced by the eye. However, overproduction of these proteins causes the formation of abnormal new blood vessels under the retina that is the hallmark of wet macular degeneration. In contrast, Lucentis, Avastin, and Macugen block only the action of VEGF but not PlGF.
View 1 and View 2 were the two pivotal Phase 3 clinical trials that proved that Eylea preserved vision (defined as loss of fewer than 3 lines on the eye chart) in 96% of treated eyes, and was equal to Lucentis, that was given to the control group of patients, in preventing loss of sight. Furthermore, patients treated with Eylea monthly gained an average of 10.9 letters on the eye chart compared to an average gain of 8.1 letters for patients treated with Lucentis; this result was highly statistically significant.
The treatment regimen recommended by the FDA for Eylea is three injections given every four weeks followed thereafter by one injection every two months. This regimen offers a significant advantage compared to Lucentis, for which the FDA recommends monthly injections. Over the course of one year, a patient would receive 7 or 8 Eylea injections compared to 12 Lucentis injections. While Eylea is costly at $1,850 per dose, it is less expensive than Lucentis, which costs approximately $2,000 per dose. The cost difference is considerable given the fewer injections of Eylea that are required for treatment. Avastin, which continues to be available as an off-label treatment for wet macular degeneration, is the least expensive medication, costing approximately $75 per dose. While the CATT trial published in April 2011 showed that Avastin and Lucentis were comparable in terms of improving and stabilizing vision in patients with wet macular degeneration, there are no studies currently available that compare Eylea to Avastin.
Obviously, at this point in time, there are as many different approaches to treating wet AMD as there are retina specialists. At The Eye Institute, we individualize the treatment to best fit the patient and one doctor will often employ different approaches depending upon the situation. Your physician may even suggest switching medications if the circumstances warrant it. Since there are so many treatment options, it is imperative for you to be well informed. Our Retina Specialists will carefully review your choices and, working together, will devise a treatment plan that will best serve your needs.
Ongoing research investigating the causes and treatment of AMD remains one of the major efforts of the ophthalmology community. Dr. Kirsch is the Principal Investigator for the evaluation of two new potential medications for wet AMD. The results of the studies should be available in the near future. The Eye Institute of West Florida is committed to providing our patients with every opportunity to benefit from the most advanced and current treatments available and will continue participating in clinical research studies in the future.
Leonard Kirsch, MD and Richard Hairston, MD were the first Retina Specialists in the Tampa Bay area to offer these new treatment options for patients with wet macular degeneration.
Schedule your Retina Evaluation today
Call (727) 581-8706 to schedule your appointment
Meet Your Retina Care Specialists
 Leonard S. Kirsch, M.D., F.R.C.S.(C) is a fellowship-trained vitreous and retina specialist. He is internationally known for the numerous papers and lectures he has presented here and abroad. Dr. Kirsch is currently an active participant in ongoing research to find new treatments for diseases of the retina. His expertise in the most advanced diagnostic and treatment techniques of all diseases of the retina, macula, and vitreous make Dr. Kirsch one of the elites in his field. In the Tampa Bay area, Dr. Kirsch pioneered the use of Photodynamic Therapy with Visudyne©, and intravitreal Macugen©, Lucentis©, and Avastin© for the treatment of age-related macular degeneration. Dr. Kirsch was also among the first surgeons in Florida to perform 25 gauge “no-stitch” vitrectomy in 2001. He is certified by the American Board of Ophthalmology and is a fellow of the Royal College of Surgeons of Canada.
Leonard S. Kirsch, M.D., F.R.C.S.(C) is a fellowship-trained vitreous and retina specialist. He is internationally known for the numerous papers and lectures he has presented here and abroad. Dr. Kirsch is currently an active participant in ongoing research to find new treatments for diseases of the retina. His expertise in the most advanced diagnostic and treatment techniques of all diseases of the retina, macula, and vitreous make Dr. Kirsch one of the elites in his field. In the Tampa Bay area, Dr. Kirsch pioneered the use of Photodynamic Therapy with Visudyne©, and intravitreal Macugen©, Lucentis©, and Avastin© for the treatment of age-related macular degeneration. Dr. Kirsch was also among the first surgeons in Florida to perform 25 gauge “no-stitch” vitrectomy in 2001. He is certified by the American Board of Ophthalmology and is a fellow of the Royal College of Surgeons of Canada.
 Richard J. Hairston, M.D. is a vitreous and retina specialist. He joined The Eye Institute in June 2001 coming to us from a busy retina practice in Washington, DC. Dr. Hairston graduated from the Johns Hopkins University School of Medicine and did his residency at the Wilmer Ophthalmological Institute at Johns Hopkins University. He completed a fellowship in diseases and surgery of the retina and vitreous at The Center for Retina Vitreous Surgery, Memphis, Tennessee. He then served as Assistant Chief of Service in Ophthalmology and Director of the Ocular Trauma Service at Johns Hopkins Hospital. Most recently he was Assistant Professor of Ophthalmology at Johns Hopkins University. He is certified by the American Board of Ophthalmology. Dr. Hairston enjoys an international reputation as an outstanding retina and vitreous surgeon.
Richard J. Hairston, M.D. is a vitreous and retina specialist. He joined The Eye Institute in June 2001 coming to us from a busy retina practice in Washington, DC. Dr. Hairston graduated from the Johns Hopkins University School of Medicine and did his residency at the Wilmer Ophthalmological Institute at Johns Hopkins University. He completed a fellowship in diseases and surgery of the retina and vitreous at The Center for Retina Vitreous Surgery, Memphis, Tennessee. He then served as Assistant Chief of Service in Ophthalmology and Director of the Ocular Trauma Service at Johns Hopkins Hospital. Most recently he was Assistant Professor of Ophthalmology at Johns Hopkins University. He is certified by the American Board of Ophthalmology. Dr. Hairston enjoys an international reputation as an outstanding retina and vitreous surgeon.
 Janie Ho M.D., is a board-certified ophthalmologist, fellowship-trained in medical and surgical vitreoretinal diseases such as macular degeneration, diabetic retinopathy and retinal tears and detachments. She has been educated at some of America’s finest institutions. She received her Bachelor of Arts from Harvard University and her medical degree from Duke University. She went on to ophthalmology residency at the University of California, San Francisco. Following residency, Dr. Ho continued on to a fellowship in vitreoretinal diseases at the prestigious Massachusetts Eye and Ear Infirmary of Harvard Medical School. Dr. Ho has participated in angiogenesis research, investigating causes and treatment for common retinal disorders.
Janie Ho M.D., is a board-certified ophthalmologist, fellowship-trained in medical and surgical vitreoretinal diseases such as macular degeneration, diabetic retinopathy and retinal tears and detachments. She has been educated at some of America’s finest institutions. She received her Bachelor of Arts from Harvard University and her medical degree from Duke University. She went on to ophthalmology residency at the University of California, San Francisco. Following residency, Dr. Ho continued on to a fellowship in vitreoretinal diseases at the prestigious Massachusetts Eye and Ear Infirmary of Harvard Medical School. Dr. Ho has participated in angiogenesis research, investigating causes and treatment for common retinal disorders.
 Sejal Shah M.D., is a board-certified, fellowship-trained Retina Specialist. Dr. Shah received her medical degree from the University of South Florida. She began her postgraduate training with an internship in medicine at UCLA, followed by ophthalmology training at the University of South Florida. She went on to complete a fellowship at the prestigious Bascom Palmer Eye Institute, specializing in the diagnosis and treatment of retinal disorders. Dr. Shah also earned a B.S. degree with honors in Nutritional Science from the University of Florida. Dr. Shah brings her expertise as a medical retinal specialist with skills in managing and treating vitreoretinal pathology which including age-related macular degeneration and diabetic retinopathy. Her research has appeared in publications such as Ophthalmic Surgery, Lasers, and Imaging Retina; Clinical Ocular Oncology; Survey of Ophthalmology; International Ophthalmology Clinics; and Survey of Ophthalmology. In her spare time, she enjoys spending quality time with her family and friends, traveling, and trying to keep up with her twin boys.
Sejal Shah M.D., is a board-certified, fellowship-trained Retina Specialist. Dr. Shah received her medical degree from the University of South Florida. She began her postgraduate training with an internship in medicine at UCLA, followed by ophthalmology training at the University of South Florida. She went on to complete a fellowship at the prestigious Bascom Palmer Eye Institute, specializing in the diagnosis and treatment of retinal disorders. Dr. Shah also earned a B.S. degree with honors in Nutritional Science from the University of Florida. Dr. Shah brings her expertise as a medical retinal specialist with skills in managing and treating vitreoretinal pathology which including age-related macular degeneration and diabetic retinopathy. Her research has appeared in publications such as Ophthalmic Surgery, Lasers, and Imaging Retina; Clinical Ocular Oncology; Survey of Ophthalmology; International Ophthalmology Clinics; and Survey of Ophthalmology. In her spare time, she enjoys spending quality time with her family and friends, traveling, and trying to keep up with her twin boys.

 Diagnostics for Macular Degeneration
Diagnostics for Macular Degeneration Leonard S. Kirsch, M.D., F.R.C.S.(C) is a fellowship-trained vitreous and retina specialist. He is internationally known for the numerous papers and lectures he has presented here and abroad. Dr. Kirsch is currently an active participant in ongoing research to find new treatments for diseases of the retina. His expertise in the most advanced diagnostic and treatment techniques of all diseases of the retina, macula, and vitreous make Dr. Kirsch one of the elites in his field. In the Tampa Bay area, Dr. Kirsch pioneered the use of Photodynamic Therapy with Visudyne©, and intravitreal Macugen©, Lucentis©, and Avastin© for the treatment of age-related macular degeneration. Dr. Kirsch was also among the first surgeons in Florida to perform 25 gauge “no-stitch” vitrectomy in 2001. He is certified by the American Board of Ophthalmology and is a fellow of the Royal College of Surgeons of Canada.
Leonard S. Kirsch, M.D., F.R.C.S.(C) is a fellowship-trained vitreous and retina specialist. He is internationally known for the numerous papers and lectures he has presented here and abroad. Dr. Kirsch is currently an active participant in ongoing research to find new treatments for diseases of the retina. His expertise in the most advanced diagnostic and treatment techniques of all diseases of the retina, macula, and vitreous make Dr. Kirsch one of the elites in his field. In the Tampa Bay area, Dr. Kirsch pioneered the use of Photodynamic Therapy with Visudyne©, and intravitreal Macugen©, Lucentis©, and Avastin© for the treatment of age-related macular degeneration. Dr. Kirsch was also among the first surgeons in Florida to perform 25 gauge “no-stitch” vitrectomy in 2001. He is certified by the American Board of Ophthalmology and is a fellow of the Royal College of Surgeons of Canada. Richard J. Hairston, M.D. is a vitreous and retina specialist. He joined The Eye Institute in June 2001 coming to us from a busy retina practice in Washington, DC. Dr. Hairston graduated from the Johns Hopkins University School of Medicine and did his residency at the Wilmer Ophthalmological Institute at Johns Hopkins University. He completed a fellowship in diseases and surgery of the retina and vitreous at The Center for Retina Vitreous Surgery, Memphis, Tennessee. He then served as Assistant Chief of Service in Ophthalmology and Director of the Ocular Trauma Service at Johns Hopkins Hospital. Most recently he was Assistant Professor of Ophthalmology at Johns Hopkins University. He is certified by the American Board of Ophthalmology. Dr. Hairston enjoys an international reputation as an outstanding retina and vitreous surgeon.
Richard J. Hairston, M.D. is a vitreous and retina specialist. He joined The Eye Institute in June 2001 coming to us from a busy retina practice in Washington, DC. Dr. Hairston graduated from the Johns Hopkins University School of Medicine and did his residency at the Wilmer Ophthalmological Institute at Johns Hopkins University. He completed a fellowship in diseases and surgery of the retina and vitreous at The Center for Retina Vitreous Surgery, Memphis, Tennessee. He then served as Assistant Chief of Service in Ophthalmology and Director of the Ocular Trauma Service at Johns Hopkins Hospital. Most recently he was Assistant Professor of Ophthalmology at Johns Hopkins University. He is certified by the American Board of Ophthalmology. Dr. Hairston enjoys an international reputation as an outstanding retina and vitreous surgeon. Janie Ho M.D., is a board-certified ophthalmologist, fellowship-trained in medical and surgical vitreoretinal diseases such as macular degeneration, diabetic retinopathy and retinal tears and detachments. She has been educated at some of America’s finest institutions. She received her Bachelor of Arts from Harvard University and her medical degree from Duke University. She went on to ophthalmology residency at the University of California, San Francisco. Following residency, Dr. Ho continued on to a fellowship in vitreoretinal diseases at the prestigious Massachusetts Eye and Ear Infirmary of Harvard Medical School. Dr. Ho has participated in angiogenesis research, investigating causes and treatment for common retinal disorders.
Janie Ho M.D., is a board-certified ophthalmologist, fellowship-trained in medical and surgical vitreoretinal diseases such as macular degeneration, diabetic retinopathy and retinal tears and detachments. She has been educated at some of America’s finest institutions. She received her Bachelor of Arts from Harvard University and her medical degree from Duke University. She went on to ophthalmology residency at the University of California, San Francisco. Following residency, Dr. Ho continued on to a fellowship in vitreoretinal diseases at the prestigious Massachusetts Eye and Ear Infirmary of Harvard Medical School. Dr. Ho has participated in angiogenesis research, investigating causes and treatment for common retinal disorders. Sejal Shah M.D., is a board-certified, fellowship-trained Retina Specialist. Dr. Shah received her medical degree from the University of South Florida. She began her postgraduate training with an internship in medicine at UCLA, followed by ophthalmology training at the University of South Florida. She went on to complete a fellowship at the prestigious Bascom Palmer Eye Institute, specializing in the diagnosis and treatment of retinal disorders. Dr. Shah also earned a B.S. degree with honors in Nutritional Science from the University of Florida. Dr. Shah brings her expertise as a medical retinal specialist with skills in managing and treating vitreoretinal pathology which including age-related macular degeneration and diabetic retinopathy. Her research has appeared in publications such as Ophthalmic Surgery, Lasers, and Imaging Retina; Clinical Ocular Oncology; Survey of Ophthalmology; International Ophthalmology Clinics; and Survey of Ophthalmology. In her spare time, she enjoys spending quality time with her family and friends, traveling, and trying to keep up with her twin boys.
Sejal Shah M.D., is a board-certified, fellowship-trained Retina Specialist. Dr. Shah received her medical degree from the University of South Florida. She began her postgraduate training with an internship in medicine at UCLA, followed by ophthalmology training at the University of South Florida. She went on to complete a fellowship at the prestigious Bascom Palmer Eye Institute, specializing in the diagnosis and treatment of retinal disorders. Dr. Shah also earned a B.S. degree with honors in Nutritional Science from the University of Florida. Dr. Shah brings her expertise as a medical retinal specialist with skills in managing and treating vitreoretinal pathology which including age-related macular degeneration and diabetic retinopathy. Her research has appeared in publications such as Ophthalmic Surgery, Lasers, and Imaging Retina; Clinical Ocular Oncology; Survey of Ophthalmology; International Ophthalmology Clinics; and Survey of Ophthalmology. In her spare time, she enjoys spending quality time with her family and friends, traveling, and trying to keep up with her twin boys.
